 Following recent mergers and acquisitions that figure is now eight of the top ten. As recently as 2005, only three of today’s top ten pharmaceutical companies had significant activities in vaccines. This situation is changing with the growth of manufacturers headquartered in developing countries and with the significant new investment by multinationals in vaccine R&D. Others, such as Merck, Pfizer and Novartis, offer a narrower range of products addressing particular disease indications or particular market niches. Among the large multinational pharmaceutical companies currently only two, Sanofi Pasteur (part of the Sanofi-Aventis group) and GlaxoSmithKline, manufacture a broad range of vaccines generally licensed for worldwide use. Although there are differences in effectiveness among vaccines, as explained elsewhere in this series (Chapter by Greenwood et al.), most have contributed significantly to the improvements in human health that we have witnessed over the past century. The actual number of vaccine products is however considerably higher, there being many combination vaccines and formulations aimed at different age groups, different geographical regions and both private and public markets. Today there are licensed vaccines available to prevent human infections caused by approximately 25 microbes. Focusing on influenza vaccines as an example that well illustrates many of the relevant points, this article considers current production, distribution, access and other factors that ultimately impact on vaccine uptake and population-level effectiveness. For vaccines whose supplies are limited, either due to rapidly emerging diseases or longer-term mismatch of supply and demand, prioritizing target groups can increase vaccine impact. 
However, ensuring optimal access and uptake also requires strong partnerships between private manufacturers, regulatory authorities and national and international public health services. Reliable vaccine production in appropriate quantities and at affordable prices is the cornerstone of developing global vaccination policies. 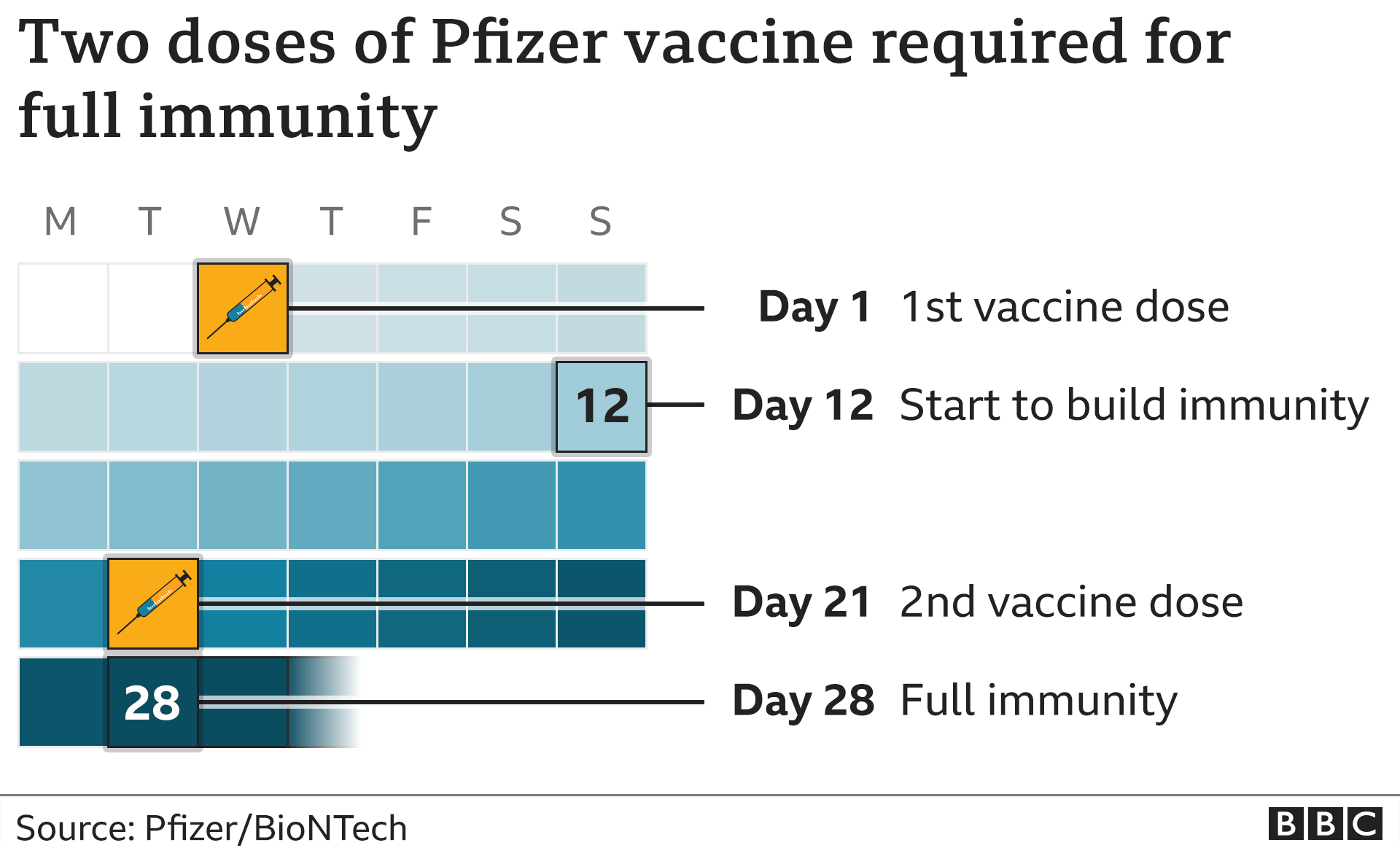
Manufacturing complexity is compounded by the need for different formulations for different countries and age groups. The technologies involved in manufacturing different types of vaccines may strongly influence vaccine cost, ease of industrial scale-up, stability and ultimately world-wide availability. Making human vaccines available on a global scale requires the use of complex production methods, meticulous quality control and reliable distribution channels that ensure the products are potent and effective at their point of use. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
